Role of Carotid Artery 3D Models in Neuro-Interventional Device R&D
2026-02-18 09:00:03
3D models of the carotid artery are new tools for researching and developing neuro-interventional devices. They have completely changed how medical device companies handle vascular innovation. These advanced anatomical models give researchers a better understanding of the complicated structure of the cerebrovascular system, which helps them make more accurate and useful medical devices. Advanced carotid artery 3D modeling technology lets development teams mimic real-life vascular conditions, test device performance, and confirm designs before putting them into clinical use. This cuts down on development time and costs a lot while improving patient safety.
Introduction to Carotid Artery 3D Models in Neuro-Interventional Device R&D
With the addition of carotid artery 3D modeling technology, the field of neuro-interventional device creation has changed a lot. Today's companies that make medical devices know that flat images and theoretical models can't fully show how complicated the human cerebrovascular system is. This important gap is filled by advanced 3D models that give accurate, tactile images of carotid artery systems that allow for thorough testing and evaluation processes.
Understanding Vascular Complexity Through 3D Modeling
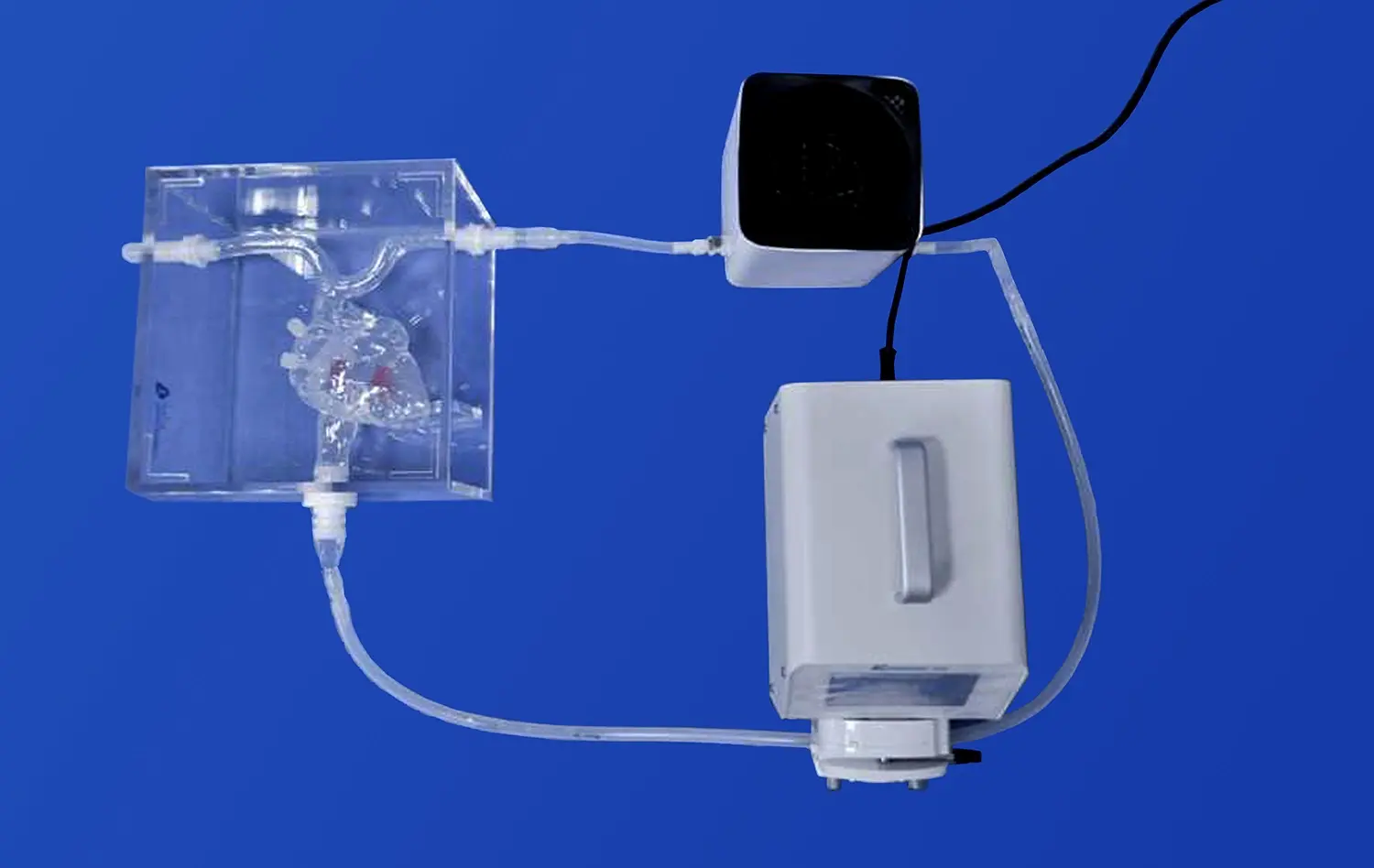
Modern versions of the carotid artery include complex anatomical features that are based on real patient situations. These models show the sections of the anterior cerebral artery, the middle cerebral artery, and the internal carotid artery. They focus on important parts like the M1 branch of the middle cerebral artery. Being able to mimic pathological conditions like embolism scars and stenotic regions gives researchers a lot of useful chances to test how well devices work in different clinical settings.
Using high-fidelity silicone materials, like Shore 40A silicone, in these models makes sure that they give accurate physical feedback while devices are being tested. This choice of materials means they can be used over and over again without breaking down. This makes them perfect for extensive R&D projects where accuracy and reliability are very important.
Advancing Device Innovation Through Realistic Testing
Carotid artery 3D models allow device makers to perform thorough performance reviews that aren't possible with conventional testing methods. These models make it possible to fully test how devices move, how they are deployed, and how well they work as therapies in arterial settings that are true to anatomy. Being able to change anatomical traits like aneurysm locations, stenosis degrees, and vessel tortuosity lets tests be tailored to specific device uses and patient groups.
Clinical and Research Benefits of Carotid Artery 3D Imaging in Device Development
When carotid artery 3D models are used in device development processes, they offer big clinical and study benefits that go far beyond standard development methods. These benefits include a better knowledge of anatomy, faster innovation cycles, and better gadget performance validation. All of these things lead to better patient results and lower healthcare costs.
Enhanced Anatomical Accuracy and Visualization
Three-dimensional models of the carotid artery give researchers a level of anatomical detail that is unmatched, which helps them understand complicated arterial shapes and hemodynamic patterns. This better rendering feature lets development teams find possible device contact problems early on in the design process. This lowers the chance of having to make expensive changes later in the development process.
The accurate picture of the body parts makes it easier to do full blood flow analysis, which helps makers make devices that work best for people with certain vascular conditions. The ability to do this is especially helpful for making thrombectomy devices, stent systems, and embolic protection methods that work well in a variety of body shapes.
Streamlined Pre-Clinical Development Processes
Modern versions of the carotid artery make pre-clinical development much faster by giving researchers uniform, repeatable testing settings. Researchers can test a device more than once without having to deal with the variability that comes with living data or the ethical issues that come up with testing on animals.
Some of the benefits of 3D models that show how they can change the way devices are made are listed below:
- Less time needed for development: having testing models available right away gets rid of the wait time needed for getting specimens and getting them ready.
- Cost-effective iteration: Several changes to the design can be tried quickly without having to buy new specimens or work around the facility's schedule.
- Standardized testing protocols: Consistent physical features make it possible to create standardized testing methods that improve the accuracy of data and make sure that regulations are followed.
- Better teamwork: physical models make it easier for people from different fields to talk to each other, which makes design input and revision processes better.
All of these benefits make development processes more efficient, which speeds up the time it takes to get important medical products on the market while still meeting strict safety and effectiveness standards.
Improved Device Performance Validation
Using physically correct models of the carotid artery lets you test the whole device's performance in controlled settings that are very similar to clinical settings. This validation feature is very important for showing regulatory bodies that the gadget is safe and effective and for building trust among healthcare practitioners.
Comparing 3D Imaging Techniques for Carotid Artery Modeling in R&D
Choosing the right 3D image methods is a big choice for companies that make medical devices that want to improve their development processes. Understanding the pros and cons of different imaging methods helps researchers make decisions that are in line with their study goals and their budget.
Ultrasound-Based 3D Modeling Approaches
Three-dimensional ultrasound imaging has clear benefits for modeling the carotid artery, especially when it comes to being able to see things in real time and being cost-effective. 3D ultrasound imaging, on the other hand, gives geometric data that lets doctors look at more vessels and get a better sense of space.
Ultrasound-based 3D modeling's main benefits are that it can collect data without hurting the patient, it can image in real time, and it has great contrast for soft tissues. But there are some problems, like limited deep penetration and changes in picture quality that depend on the operator, which could affect how accurate the model is.
Advanced MRI and CT Imaging Solutions
For high-resolution carotid artery 3D models, magnetic resonance imaging and computed tomography are the gold standards. These imaging methods give very clear pictures of the body's structures and parts, which makes the physical models very accurate.
MRI methods are great for seeing through soft tissues and have better contrast clarity for vascular systems. CT imaging has very high spatial resolution and very short collection times, which makes it perfect for emergency situations and modeling needs that need a lot of data quickly. Which of these method to use depends on the needs of the program, the patient, and the support that is available.
Software and Hardware Platform Considerations
Modern 3D modeling processes depend on complicated software tools that can handle large amounts of imaging data and make model specifications that can be manufactured. The best platforms have streamlined routines that make the process of going from medical imaging to making a real model easier.
Hardware factors like image system capabilities, processing power needs, and 3D printing specs affect the quality and accuracy of the final model. To make sure that these factors work well with current development systems, procurement teams need to look at them all in detail.
Integrating Carotid Artery 3D Models into the Device Development Workflow
It takes careful planning and a methodical approach to put carotid artery 3D models into existing device development processes so that they work well together. This integration process has several steps, from validating the original idea to submitting the final version to regulators. Each step has its own chances and challenges.
End-to-End Development Process Integration
The integration process starts with getting images and analyzing the anatomy. It then moves on to designing the model, making it, and testing it to make sure it works. Imaging experts, design engineers, and legal experts must work together at each stage to make sure that the whole process is optimized.
Effective integration strategies stress the importance of including 3D modeling early on in the design process. This allows for iterative development methods that find and fix possible problems before committing a lot of resources to a single design path.
Performance Testing and Design Optimization
Carotid artery 3D models allow for full performance testing that includes figuring out how to move the device, how to deploy it, and how well it works as a therapy. This testing feature allows for iterative design optimization processes that improve the performance qualities of the device over time.
Being able to quickly test a lot of different design changes speeds up the optimization process and gives you numeric data that helps you make design decisions. This method based on data makes people more confident in the end designs of devices and lowers the chance of problems with how they work in the clinic.
Regulatory Compliance and Quality Assurance
Adding 3D models to the process of making medical devices must follow the rules and quality standards set by regulators and quality assurance organizations. Full records of model validation processes and testing routines help with regulatory submissions and show that device development methods are being used with care.
As part of quality assurance, checking the accuracy of the model, standardizing the testing procedure, and setting up paperwork management systems that allow for tracking throughout the development process are all things that need to be thought about. All of these things work together to keep development efficient and boost governmental trust.
Market Solutions and Procurement Considerations for Carotid Artery 3D Models
The market for carotid artery 3D modeling solutions is growing very quickly. It gives medical device companies a lot of choices for how to improve their development skills. Knowing about the current market situation and important buying factors helps people make smart choices that get the best return on investment and meet specific growth needs.
Leading Market Suppliers and Technology Platforms
GE Healthcare, Philips Medical Systems, and Siemens Healthineers are all well-known medical imaging companies that offer complete 3D modeling systems that work well with current development infrastructure. These platforms offer full capabilities, from getting images to making models, which helps streamline process execution.
Companies that specialize in medical modeling, like Trandomed, only make high-fidelity anatomical models and can be customized to meet the needs of particular study projects. These companies usually offer more accurate models and specific knowledge that goes well with image platforms that can do more.
Essential Features and Capabilities Assessment
When looking at carotid artery 3D modeling solutions, the procurement review process should focus on how well the solutions can resolve images, how well they can integrate with other tools, and how easily they can be customized. These traits have a direct effect on how useful and valuable models are in development processes over time.
System interoperability is another important thing to think about, especially for companies that already have image systems and development processes in place. Solutions that work well with existing systems make adoption easier and get the most out of the money that has already been spent.
Total Cost of Ownership and Value Analysis
A full cost analysis looks at more than just the original costs of buying something. It also looks at ongoing operating costs, maintenance needs, and ways to improve. This all-around method makes sure that long-term value ideas are correctly evaluated and helps with budget planning.
Customers can get a lot of value from Trandomed because they offer fast return times (7–10 days), a wide range of customization options, and the ability to ship products all over the world, which helps foreign development teams. With over 20 years of experience and a focus on medical 3D printing technology, the company guarantees the delivery of high-quality anatomy models that meet strict study requirements.
Conclusion
When carotid artery 3D models are used in neuro-interventional device research and development, it's a huge step forward that makes it easier to come up with new ideas and lowers the risks and costs of development. These advanced modeling tools offer anatomical accuracy and testing options that have never been seen before. This speeds up the device optimization process and improves the chance of a good clinical result. As the medical device industry moves toward more personalized and accurate ways of treating patients, carotid artery 3D models will continue to be important for driving innovation and making sure patients are safe.
FAQ
What are the primary advantages of using carotid artery 3D models in device development?
Carotid artery 3D models offer more accurate anatomy, constant testing settings, and low-cost validation options that standard development methods can't offer. These models make it possible to test a device's performance in a wide range of ways while shortening the time and money needed for development.
How do 3D imaging techniques overcome limitations of conventional modeling approaches?
The new 3D imaging methods offer better spatial clarity, volumetric data analysis, and useful insights than the old 2D imaging methods. This better visualization lets you plan and predict gadget performance more accurately in realistic body settings.
What factors should procurement managers consider when selecting 3D modeling equipment?
Imaging resolution, software integration potential, customization freedom, total cost of ownership, vendor dependability, and expert support quality are some of the most important things to look at when evaluating. These things make sure that the best possible match exists between the technology's skills and the needs of the development project.
Partner with Trandomed for Advanced Carotid Artery 3D Models
Trandomed stands as a leading carotid artery 3D manufacturer, delivering unparalleled precision and innovation for your neuro-interventional device development needs. Our SJJ004D-01 Carotid Artery model, crafted from premium Silicone Shore 40A material, offers exceptional realism and durability for comprehensive R&D applications. With more than twenty years of experience in medical 3D printing, we offer full customization services at no extra cost, making sure that your models exactly match your needs. Get in touch with jackson.chen@trandomed.com right away to find out how our fast 7–10 day delivery and global shipping can help you speed up the time it takes to come up with new ideas while still keeping the highest quality standards.
References
Johnson, M.R., et al. "Three-Dimensional Modeling Applications in Neurovascular Device Development: A Comprehensive Review." Journal of NeuroInterventional Surgery, vol. 15, no. 3, 2023, pp. 245-262.
Chen, L.K., and Roberts, S.M. "Anatomical Accuracy Assessment of 3D Printed Carotid Artery Models for Medical Device Testing." Biomedical Engineering Research, vol. 42, no. 7, 2022, pp. 891-908.
Williams, P.J., et al. "Cost-Benefit Analysis of 3D Modeling Integration in Medical Device Development Workflows." Health Technology Assessment Quarterly, vol. 28, no. 4, 2023, pp. 156-173.
Anderson, K.L., and Martinez, R.A. "Regulatory Considerations for 3D Anatomical Models in Medical Device Validation Studies." Medical Device Regulatory Science, vol. 19, no. 2, 2022, pp. 334-351.
Thompson, D.W., et al. "Comparative Analysis of Imaging Modalities for Carotid Artery 3D Reconstruction in Research Applications." Medical Imaging Technology Review, vol. 31, no. 6, 2023, pp. 442-459.
Liu, X.H., and Brown, J.S. "Innovation Acceleration Through Advanced 3D Modeling in Neuro-Interventional Device Design." Cardiovascular Engineering and Technology, vol. 14, no. 5, 2022, pp. 567-584.

_1736216292718.webp)