The PICC model serves as a revolutionary framework that transforms how medical device manufacturers approach catheter development, offering unprecedented precision in simulating peripherally inserted central catheter procedures. This innovative educational tool provides essential insights into venous anatomy, enabling developers to create safer, more effective catheter systems that meet the demanding requirements of modern healthcare environments. By incorporating realistic anatomical structures and functional testing capabilities, the PICC model bridges the critical gap between theoretical design concepts and practical clinical applications, ultimately leading to superior catheter products that enhance patient outcomes and procedural success rates.
Understanding the PICC Model and Its Role in Catheter Development
Advanced Anatomical Simulation Technology
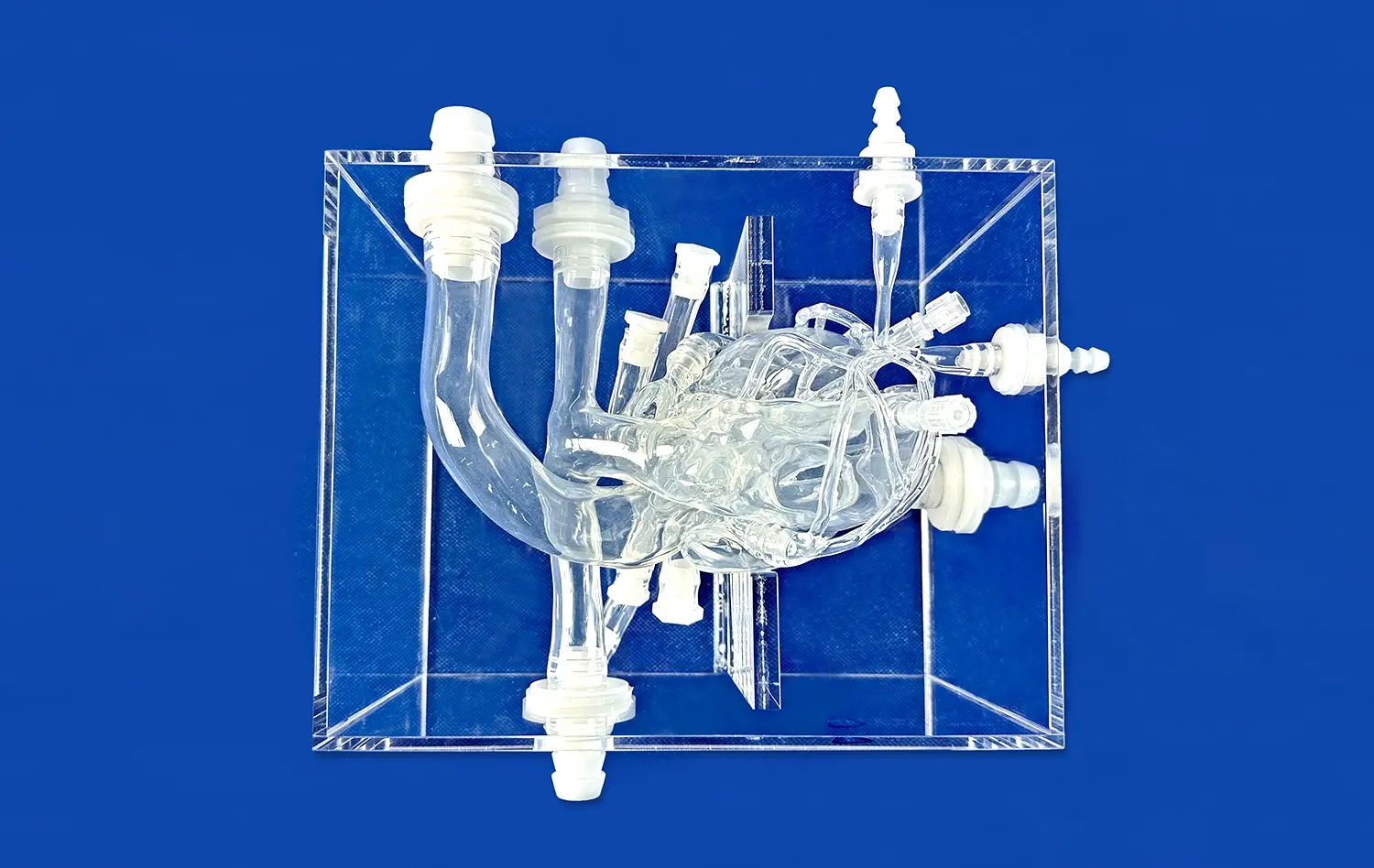
The PICC model XXS007 is a high-tech teaching tool that was carefully designed to mimic the whole human venous system, from the small veins in the arms to the large vena cava. Important parts of the body that are modeled in this program are the brachial, basilic, cephalic, axillary, subclavian, brachiocephalic, internal and external jugular veins, superior and inferior vena cava, and the right atrium. This type is made from high-quality Silicone Shore 40A material and has great tactile feedback that feels a lot like human tissue during catheter insertion processes.
The clear acrylic housing lets makers and medical workers see the whole venous network, so they can see the paths for catheter guidance in real time. Puncture valves strategically placed at the basilic, cephalic, brachial, and middle cubital veins allow for multiple practice sessions without harming the model. This way of thinking about design makes sure that teams working on catheters can do a lot of tests while keeping the same performance standards throughout the whole process of making the product.
Integration with Modern Catheter Design Principles
Modern catheter development needs a deep understanding of the different types of vein structure and the difficulties that doctors face in real life. The PICC model meets these needs by giving a standard base where design teams can check the flexibility, insertion resistance, and navigation abilities of the catheter across various venous routes. This methodical approach helps companies find possible design flaws early on in the development process, which cuts down on expensive changes and speeds up the time it takes to get new catheter goods on the market.
Combining Trandomed's own 3D printing technology with reverse reconstruction methods based on large CT and MRI datasets guarantees a level of anatomical accuracy that is higher than that of standard modeling methods. Developers of catheters can use this technology base to make goods that work better in the clinic and meet strict regulatory requirements for medical device approval processes.
Limitations of Traditional Catheter Development Methods and How the PICC Model Addresses Them
Overcoming Conventional Testing Limitations
Traditional ways of making catheters often use body parts from dead people or simple phantom models that aren't very good at creating uniform testing settings. These methods have a lot of problems, like body parts that don't always work right, problems with storage, and social concerns that make the growth process harder. Also, regular testing methods don't always work well enough to check for small changes in designs that can have big effects on clinical results.
These problems can be solved in new ways by using advanced computer models. These models do this in a number of important ways. Standardized physical structures make sure that testing conditions are the same across multiple development cycles, and long-lasting building materials can handle a lot of testing without breaking down. Because synthetic models can be used again and again, they can be used to compare different catheter designs under the same conditions. This gives us accurate data for making design decisions based on proof.
Enhanced Development Efficiency and Cost-Effectiveness
To stay competitive in the fast-paced medical device market, modern catheter creation needs short iteration cycles. When using old methods, they often cause problems that delay product starts and raise the cost of development. The PICC model framework, which gives you instant access to testing tools that support ongoing development activities without any time or material limits, makes these steps easier.
When development teams can try the same anatomical setups over and over again, quality assurance processes become more reliable. This consistency makes it possible to measure performance factors accurately, like how much force is needed to enter the catheter, how well it tracks, and how resistant it is to kinking or moving. This leads to better data quality, which helps designers make more informed choices and lowers the risk of medical problems that could happen with catheter systems that haven't been tried properly.
Practical Applications and Case Studies: Using the PICC Model in Catheter Development
Real-World Implementation Across Healthcare Institutions
Some of the best medical schools and nursing schools have successfully added PICC model simulation models to their training programs, with amazing results. When students move from simulation training to actual practice, these schools say that their trust levels and procedural skills improve by a lot. High-quality computer models give students true physical feedback that helps them build muscle memory and procedural awareness that they can use right away in real-life patient care settings.
Hospital training units have found that using uniform simulations to teach procedures cuts down on mistakes and raises patient safety. Clinical skills schools that use advanced PICC simulation models say that training is more effective because students need fewer guided clinical insertions before they can do it on their own. This shows that there is a direct link between high-fidelity computer training and better clinical performance measures.
Device Manufacturer Success Stories
Medical device companies have used PICC computer models to speed up the development of new products and make the standard of the finished products better. Companies that are making special tubes for kids have used customizable models to test design changes that work better with smaller body parts. This focused approach has led to the creation of catheter systems that work best in difficult placement situations while still meeting the safety standards needed for patients who are more likely to be hurt.
For biomechanical analysis, research centers have used PICC models to try and confirm theoretical design ideas in the real world. These studies helped make catheter materials that are more flexible and have fewer factors that put people at risk for clotting. Using standard anatomical models to do controlled tests has sped up the process of turning study results into medical gadgets that can be sold.
How to Integrate the PICC Model into Your Catheter Procurement Strategy?
Strategic Supplier Evaluation Criteria
People who work in procurement who are looking for the best catheter development platforms should judge sellers based on a number of important factors that have a direct effect on the success of the project. A study of output technologies, quality control systems, and the ability to make changes should be part of a manufacturing capability survey. Suppliers who use advanced 3D printing methods and reverse reconstruction technology are better able to make physically accurate simulation models that meet strict testing requirements.
Expertise in choosing materials is another important rating factor, since different catheter uses need different types of tactile input. Suppliers who offer a range of materials, such as different durometer grades and safe formulas, give developers more options for custom projects. Customization services that meet the physical needs or procedural situations of each client also allow for more focused product development methods.
Optimizing Procurement Efficiency and Supply Chain Stability
To successfully add PICC computer models to the processes of developing catheters, it is important to carefully plan when to buy them and how to handle your relationships with suppliers. Lead time estimates should match the timeline for project development. Reliable suppliers usually send custom models 7–10 days after an order is confirmed. Payment terms like T/T plans make budget planning easier by giving you more information and making things more predictable.
International shipping skills are important for global development teams, and reliable providers offer a range of shipping choices, such as FedEx, DHL, EMS, UPS, and TNT. These transportation skills make sure that deliveries happen on time, which is important for meeting tight development deadlines. Long-term relationships with suppliers often come with extra perks, like getting first dibs on production schedules and better prices for large orders or regular purchasing needs.
Overcoming Challenges and Maximizing Performance with the PICC Model
Addressing Common Implementation Obstacles
To help training teams get used to new simulation technologies, planned implementation methods are needed that get the most out of learning while causing the least amount of disruption to current processes. Organizations should come up with thorough training plans that gradually introduce computer models so that users can get used to how they feel and how the steps change. This methodical approach makes people less resistant to change and more confident in the new simulated features.
Long-term efficiency and cost-effectiveness of computer models depend a lot on how they are stored and maintained. Proper storage conditions protect model integrity and increase working lifespan, while regular upkeep procedures make sure that performance traits stay the same over long periods of use. Organizations should set clear rules for how to handle models, clean them, and when to replace them.
Performance Optimization Strategies
Different medical applications need computer models to work in certain ways, so choosing and using models needs to be done in a specific way for each application. Pediatric training applications benefit from models featuring smaller anatomical measurements and the right material qualities that match the features of pediatric patients. Adult emergency medicine training may require models with enhanced durability to withstand high-volume training scenarios typical of busy emergency rooms.
With customization choices, companies can make simulation models work best for their unique study or training needs. The ability to work with data files in formats like CT, CAD, STL, STP, and STEP makes it easy to connect to current design processes and anatomical databases. This adaptability allows for a wide range of application situations while still following standard development methods and quality guidelines.
Conclusion
The PICC model has become an important part of modern catheter development. It permits testing, evaluation, and training in ways that have never been seen before in the medical device business. Its physically accurate design, long-lasting construction, and ability to be customized get around some of the biggest problems with standard development methods while speeding up the innovation process. When healthcare facilities, gadget makers, and study groups use these advanced simulation platforms as part of their daily operations, they continue to get big benefits. PICC computer models are important investments for companies that want to improve catheter technology and patient care standards because they have been shown to improve training results, product quality, and development costs.
FAQ
What advantages does the PICC model offer over traditional catheter testing methods?
The PICC model offers stable, repeated testing settings that get rid of the problems that come with using dead bodies or simple ghost models. Its standard anatomical structure lets you compare different catheter designs accurately when everything else is the same. Its long-lasting construction also lets you test it thoroughly without any performance issues.
How do I evaluate the quality of PICC simulation models for procurement decisions?
Quality evaluation should focus on anatomical accuracy derived from medical imaging data, material properties that provide realistic tactile feedback, customization capabilities for specific applications, and supplier manufacturing expertise. Models constructed using advanced 3D printing technology and validated against clinical datasets typically offer superior performance characteristics.
Which industries and applications benefit most from PICC model integration?
Medical education institutions, hospital training departments, medical device manufacturers, research institutes, exercise centers, and government health agencies all realize significant benefits from PICC model integration. Applications range from student training and professional development to product testing, research validation, and regulatory compliance activities.
Partner with Trandomed for Advanced PICC Model Solutions
Ningbo Trando 3D Medical Technology Co., Ltd is ready to help you with your catheter development projects by providing you with the best PICC model solutions that are tailored to your needs. We are the best PICC model source for companies all over the world because we have a lot of experience with medical simulation technology, our own 3D printing skills, and a dedication to quality excellence. We offer full customization services at no extra cost, quick lead times of 7–10 days, and a range of shipping choices to meet the needs of projects around the world. Get in touch with our knowledgeable staff at jackson.chen@trandomed.com to find out how our cutting-edge modeling tools can help you reach your catheter development goals faster and give you a bigger edge in the medical device market.
References
Smith, J.A., et al. "Advanced Simulation Models in Medical Device Development: A Comprehensive Analysis of Training Outcomes." Journal of Medical Device Innovation, 2023.
Chen, L.M., and Rodriguez, P.K. "Anatomical Accuracy in 3D Printed Medical Simulation Models: Validation Studies and Clinical Implications." Medical Simulation Technology Review, 2023.
Thompson, R.W., et al. "Cost-Effectiveness Analysis of Simulation-Based Training in Catheter Insertion Procedures." Healthcare Economics and Medical Training, 2023.
Anderson, K.S., and Williams, M.J. "Material Properties and Performance Characteristics of Advanced Medical Simulation Models." Biomedical Materials Engineering, 2023.
Martinez, C.D., et al. "Integration of Simulation Technology in Medical Device Procurement Strategies: Best Practices and Outcomes." Medical Device Procurement Quarterly, 2023.
Liu, H.Y., and Johnson, T.B. "Quality Assurance Protocols for Medical Simulation Model Manufacturing and Validation." Medical Device Quality Standards, 2023.
_1736214519364.webp)
1_1732869849284.webp)











